The Food Safety and Standards Authority of India (FSSAI) recently approved amendments to food safety regulations, eliminating the need for Bureau of Indian Standards (BIS) or Agmark certifications for food products. The move aligns with the “One Nation, One Commodity, One Regulator” concept, streamlining certification processes for food businesses and fostering a more business-friendly environment. Discussed during the 43rd meeting of the food safety authority in New Delhi, the amendments aim to simplify regulations. Additionally, a comprehensive manual of methods of analysis for ensuring regulatory compliance of food products was approved during the meeting.
This step marks progress in concentrating a Food Business operator’s focus on a single agency, freeing them from multiple differential standards for certain products. Although BIS plays a crucial role in developing standards for various critical products, the recent amendments by FSSAI intend to consolidate certifications, making FSSAI certification the sole requirement. Until the amendments are approved, the dairy industry currently requires separate BIS certification for certain products, like dried milk categories. Infant milk powder, a critical product in this category, follows stringent standards, and Indian infant milk products are considered among the safest globally. As the name suggest, this product is used by the most vulnerable group of infants and toddlers.
BIS became the first certification body in the world to reduce the SPC count in finished product to below 500/gm in three samples out of five as a testing protocol. The other countries which stood very near to India are GCC nations at 1000 CFU/g and CIS countries at 2000 CFU/g.
Is it true that Indian infant milk powder is safest in the world ?
The answer is both yes and no. When we look at world’s best names in this category then we have Nestle, Danone, Abbot, Amul, Nutricia, Nutrimed etc already manufacturing their products in India. Infant milk powder manufacturing is a complex process requiring hygiene and food safety comparable to that of pharmaceutical sector. These companies have set up state of the art technology plant and machineries to manufacture the product matching international standards.
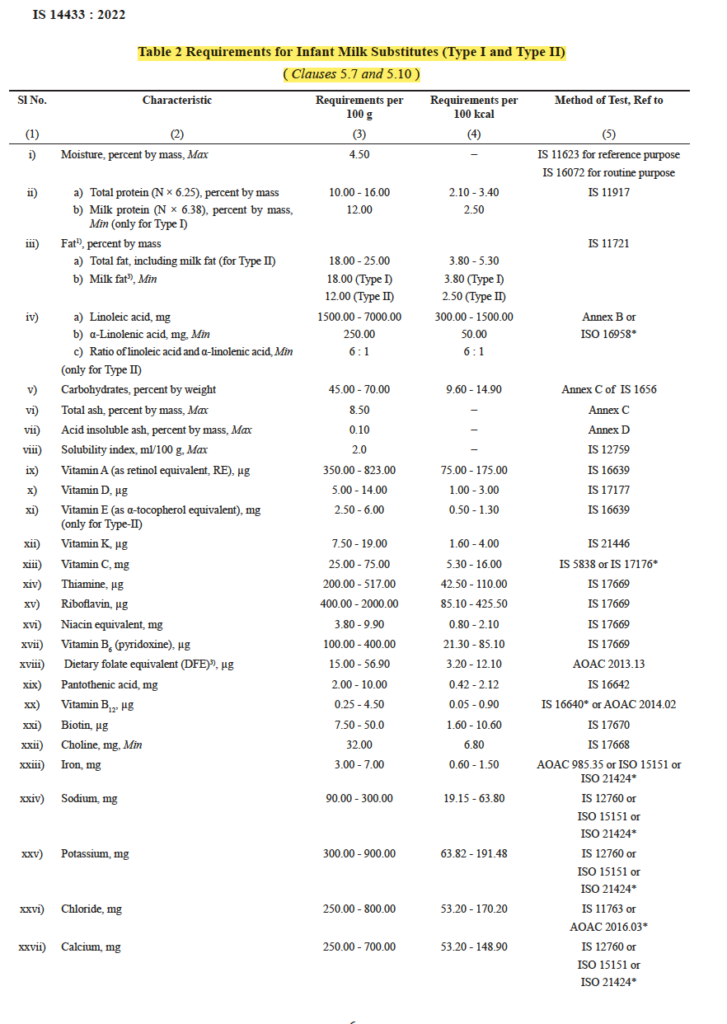
The standards for infant milk substitutes, outlined in BIS standards under IS 14433:2022, emphasize nutritional suitability and quality criteria.
“A breast milk substitute product based on milk (see 4 of IS 13688) and other ingredients which has been proven to be suitable for infant feeding, to meet the nutritional requirements of infant during the first six months. The product may be modified by the partial removal/substitution of milk fat with edible vegetable oils rich in polyunsaturated fatty acids and/ or by different milk solids, either singly or in a suitable combination; carbohydrates; salts such as phosphates and citrates; vitamins and minerals. The product may be in liquid or powdered form. Vegetable oils rich in polyunsaturated fatty acids may be added to partially substitute milk fat to an extent that the product shall contain a minimum of 12 percent by mass of milk fat.”
IS 14433 :2022 Infant milk Formula
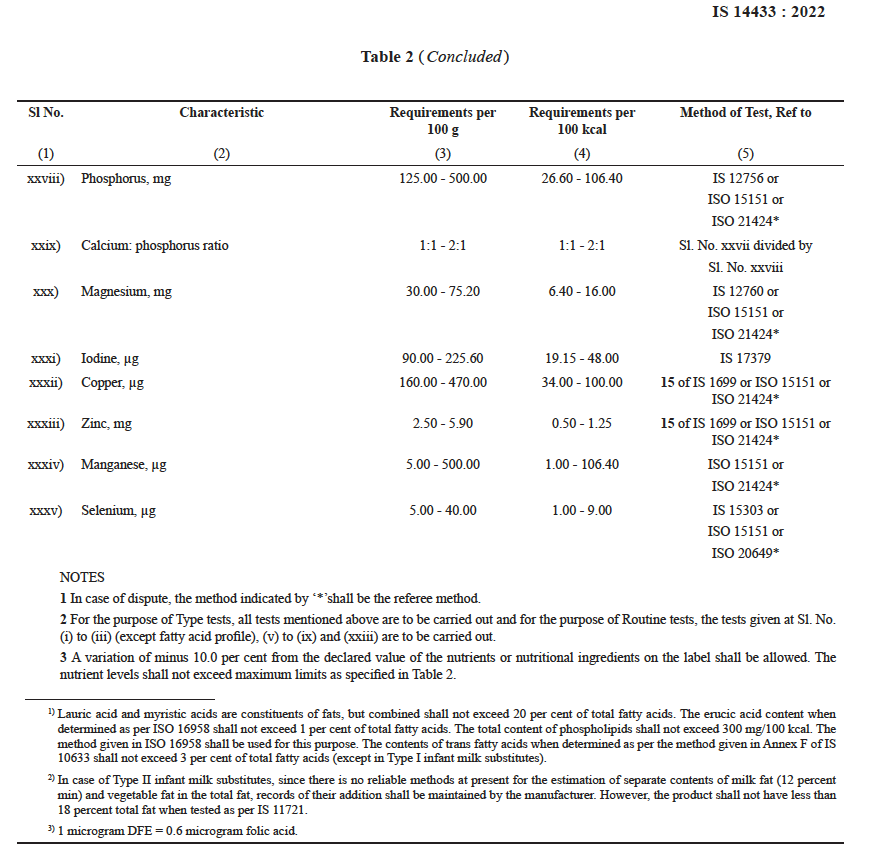
The final requirements of the product is prescribed as below :
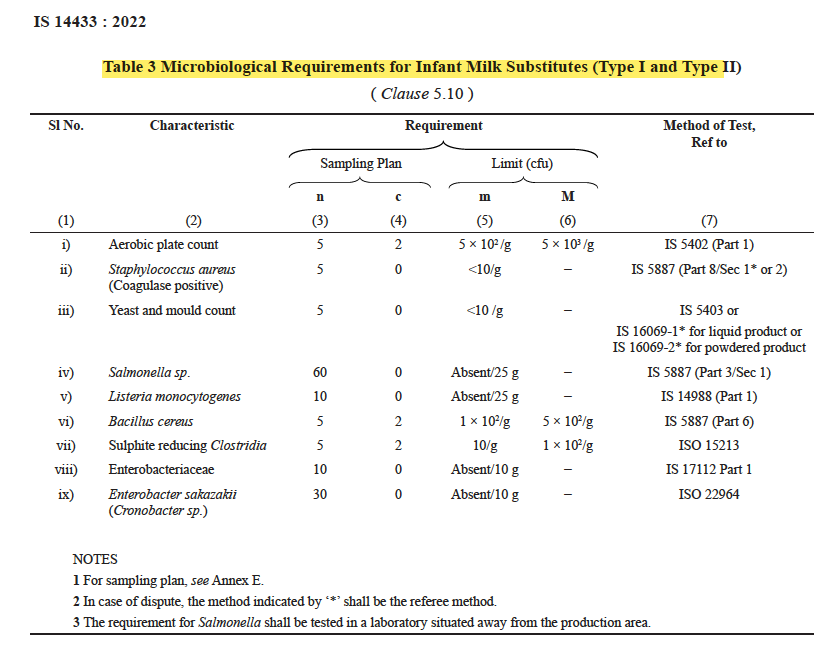
This product must match the following microbial criteria which is probably most stringent in the world from SPC perspective :
You might be wondering that why I am sharing all these details here while everyone know about it. In order to make things more simpler for you, let me share the excerpts from Skimmed milk powder also under BIS
IS 13334 (Part 1) : 2014.Skimmed Milk Powder
Skimmed Milk Powder, Standard Grade —The product obtained from skimmed milk of cow or buffalo or a combination thereof by the removal of water through spray drying or by roller drying.
We can clearly see that the process by which the milk is to be dried is clearly defined. Skimmed milk powder is a commoditised product but for that very clear instruction on its method of manufacturing is provided. Even BIS certification may not be given to an entity which does not have their captive milk pasteurising, condensing and drying facilities.
In contrast, the processing methodology for the most delicate dairy product, specifically designed for the vulnerable demographic of infants and toddlers, lacks a defined procedure. The sole requirement is to ensure that the product aligns with the previously mentioned standards and maintains a microbial count of SPC 500/gm. Manufacturers are only obligated to have a microbiological testing laboratory, which can be established at a relatively modest cost, typically a few lakhs. Notably, there is no applicable standard, such as ISO 17025, governing the established laboratory in this context.
How can we develop a product with a microbial count of less than 500/gm when the ingredients have a significantly higher microbial load, and all of this without employing any thermal processing methods?
The ingredients in infant formula possess varying densities. When these are dry-blended and pillow-pouched with nitrogen flushing for transportation, heavier ingredients such as sugar tend to settle down. This results in the product becoming a multilayered formula instead of a uniformly blended composite formula. Consequently, when served to infants, the scoops at the top will be lower in sweetness, while the last scoops will be excessively sweet. Infants are unable to communicate their discomfort, and the intended purpose of each serving with similar nutritional composition is undermined.
The remedy
The only remedy is to make wet blending of ingredients mandatory for infant milk products because only wet blending and drying could ensure SPC count of 100-500 cfu/gm . Wet blending followed by drying also ensure high solubility and no chance of segregation of ingredients and thus ensure a homogeneous product in every scoop.
I urge the FSSAI and BIS authorities to thoroughly examine and establish clear standards for Infant milk formula processing. The presence of numerous small-scale operators in this sector poses a significant threat to the health of infants in the country. Policymakers and authorities should conduct surveillance at hospitals to assess the prevalence of medical cases involving vomiting and diarrhea in infants who have been fed with such infant formulas. This step is crucial for ensuring the well-being and safety of the vulnerable infant population.